Healing Damaged Joints with Regenerative Medicine
Within the RegMed XB consortium, researchers are developing solutions to repair joint defects, primarily in the knee, through regenerative medicine. These efforts are carried out within the Osteoarthritis (OA) Moonshot and supported by the broader infrastructure of the RegMed XB Pilot Factory, one of the largest international infrastructures for regenerative medicine.
In this interview, Frank Luyten, Medical Scientific Director of RegMed XB and Professor Emeritus of Rheumatology at KU Leuven/UZ Leuven, and Maaike Braham, Regenerative Medicine and Tissue Engineering Specialist at the Innovation Center for Advanced Therapies (ICAT), one of the five pilot lines within the RegMed XB Pilot Factory, discuss future possibilities for repairing damaged joint surfaces, cartilage, and underlying bone using regenerative medicine.
Published during Rheumatology Week 2026, the interview highlights how advanced therapy development and pilot manufacturing bring regenerative medicine innovations to patients.
-
RegMed XB Pilot Factory
Funded by Dutch National Growth Fund (Nationaal Groeifonds), the RegMed XB Pilot Factory supports the Osteoarthritis (OA) Moonshot in the development of translational regenerative medicine products.
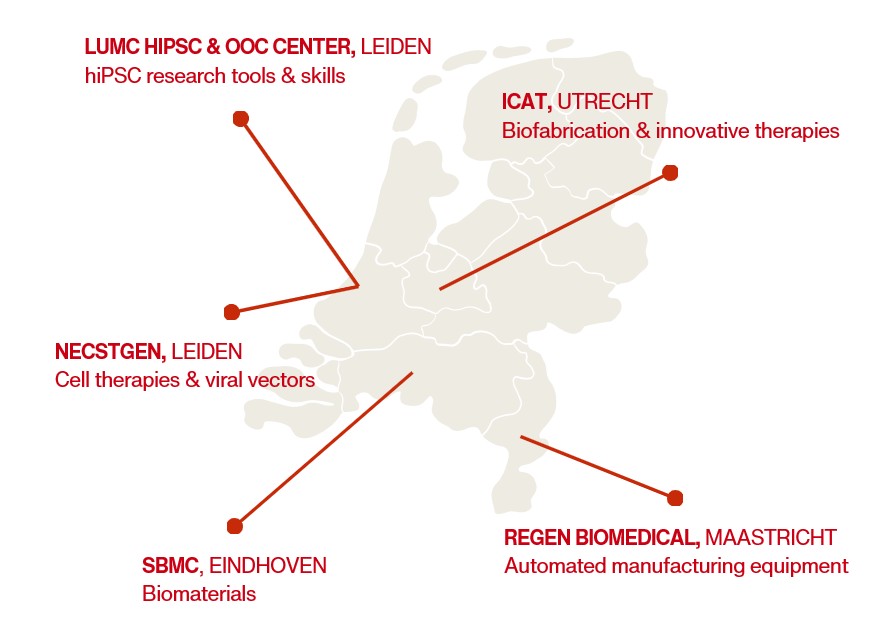
The RegMed XB Pilot Factory consists of five pilot lines, each with its own specialization:
- Innovation Center for Advanced Therapies (ICAT): bridges academic research and clinical application by integrating expertise and infrastructure for early-stage development, GMP simulation, and patient-specific ATMP production.
- Leiden University Medical Center (LUMC): a center of expertise to improve efficiency, quality and valorization of in vitro modeling technologies and human induced pluripotent stem cell research-grade products, by providing services and training.
- NecstGen: provides ATMP process development and GMP manufacturing services from preclinical development through to Phase III clinical trials.
- ReGEN Biomedical: exploits and develops scalable and automated manufacturing equipment to ensure consistency, quality, and industrial feasibility in cell and tissue production. ReGEN also enables semi-automated cartilage manufacturing
- Smart BioMaterials Center (SBMC): provides biomaterials R&D and regulatory-aligned testing, combining advanced technology with indepth analytical support for biocompatibility, cell-materials interactions and material characterization to bridge early R&D and GMP-compliant clinical manufacturing of biomaterial-based therapies.
Osteoarthritis Moonshot
Professor Luyten focuses on isolated joint surface injuries. "With this type of injury, there is a localized 'wound' in the cartilage and often also the underlying bone, but the joint itself is still healthy. This mainly occurs in young people due to trauma such as sports injuries or traffic accidents. For this, the joint must only show mild signs of osteoarthritis. It often involves cartilage injuries that extend into the underlying bone. Subsequent activities place additional load on the joint."
If left untreated, the risk of developing a diseased arthritic joint increases, especially if the injuries extend deep into the underlying bone.
"Cartilage cannot heal like a broken bone; the wound can only partially heal. The scar tissue formed is not as good as the original cartilage. The pain often returns due to mechanical loading." So far, Professor Luyten has not seen a robust solution capable of fully restoring the original tissue. “Unfortunately, current treatments and products do not significantly reduce the risk of developing osteoarthritis.” Preventing this progression is exactly what Professor Luyten aims to achieve with regenerative medicine. “Up to half of all patients could benefit from this.”
Current possibilities
Professor Luyten indicates that currently, cell therapy (using the patient's own cells) can significantly restore cartilage, at least in some patients. "For this, we use small amounts of the patient's cartilage cells, which we multiply by culturing them in a laboratory. This way, newly generated cartilage can be implanted in the patient." Unfortunately, this rather expensive solution is not always successful, because the patient must meet several clinical criteria. "This treatment often works best in young patients with recent injuries, where the rest of the joint is still in good condition."
Moreover, this treatment requires a great deal of patience. Only after six weeks can the joint be subjected to limited weight-bearing, allowing the effectiveness of the treatment to be assessed. After twelve weeks, the joint can be fully weight-bearing. Intense sports are not possible for several months. Patience is worth it, assures Luyten: "There is a good chance of full recovery for some of them, and therefore no more cartilage problems."
Future possibilities
“One of the reasons for the limited results is that in many patients with clear and chronic symptoms such as pain during weight-bearing, the underlying bone is also involved and damaged. These are precisely the patients who have a higher risk of developing osteoarthritis. That is why RegMed XB focuses on this patient group, and we are also trying to restore the cartilage-bone interface. This is a step further but does lay the foundation for a solution for early osteoarthritis and osteoarthritis prevention. To address this, fortunately there are many new possibilities on the horizon, such as breakthroughs in stem cell therapy and new biocompatible biomaterials.” says Frank Luyten.
This scientific breakthrough allows adult cells to be reprogrammed into stem cells. The Japanese scientist Shinya Yamanaka won the Nobel Prize in Physiology or Medicine in 2012 for this.
Luyten continues: "Stem cells, without limitations in number, can create tissues, such as bones, kidneys, liver and therefore also cartilage and joints. This breakthrough is currently being further researched and developed for many applications, including joint repair."
In addition, a new technology has been developed in which, starting from stem cells, mini tissues, also called organoids, can be made on a large scale in the lab. In Leuven (RegMed XB - Flanders), cartilage and bone-forming organoids have been scaled up using the Prometheus platform in a fully automated production platform, co-developed with Fraunhofer IPT in Aachen. These serve as building blocks to be assembled into larger implants tailored to the individual patient via 3D bioprinting.
3D Bioprinting
Maaike Braham, Regenerative Medicine and Tissue Engineering Specialist at the Innovation Center for Advanced Therapies (ICAT), one of the RegMed XB Pilot Lines, explains: 3D bioprinting of living tissues. With this technology, researchers are developing new regenerative therapies that can repair damaged tissue and, through ICAT, should ultimately become available to patients more quickly.
“At ICAT, we are developing new treatments to repair damaged tissues, such as cartilage, bone, and heart tissue,” says Braham.
“We use 3D bioprinting to incorporate living cells into specialized materials (bioinks) and position them precisely where they are needed. We can also print supporting structures that give the new tissue strength and help cells grow in the right direction.”
This can be important, for example, in the case of joint damage. “If not only the cartilage but also the underlying bone in a damaged joint is affected, this technology can make a significant difference. In principle, we can create a custom piece of cartilage and bone composed of living tissue that perfectly fits the patient’s damaged joint. In the future, this could mean that the joint can remain intact and that a plastic or metal prosthesis will no longer always be necessary.”
ICAT brings together all the necessary expertise and infrastructure to develop and optimize innovative therapies, ensuring they reach patients faster. However, important steps are still needed before these treatments are widely available.
“We work with living cells and advanced materials, so the production of these therapies is highly complex. We also need to ensure consistent quality and demonstrate that the technology can ultimately be applied at scale and at an affordable cost.”
According to Braham, development is moving quickly. “Our current goal is to start the first clinical studies within approximately five years.”

Background information on the Osteoarthritis (OA) Moonshot
The RegMed XB Osteoarthritis Moonshot is a large public-private research programme in regenerative medicine. The programme receives funding through the Public–Private Partnership (PPS) programme of Health~Holland. The goal is to develop biological solutions for osteoarthritis, so that damaged joints no longer need to be replaced with artificial prostheses but can be biologically repaired.
This solution is aimed at patients under 60 years of age. For this group, an artificial prosthesis is less ideal, as the lifespan of an artificial prosthesis is approximately 20 years and people are living longer.
A first generation of implants is intended to repair defects in the knee joint up to a surface area of 4 cm², including damage that extends into the underlying bone. These implants have a cartilage component containing living cells and a bone anchor without cells that supports bone ingrowth (osteoconductive).
The second generation of implants will be entirely "living" implants made with stem cells and aim to replace partial or complete joints, again for younger patients.
Involved knowledge institutions, industry, and companies
The Moonshot is part of a larger ecosystem of research clusters in the Netherlands and Flanders, including Leiden University Medical Center, UMC Utrecht, Utrecht University, Eindhoven University of Technology, Maastricht University, and Scinus Cell Expansion. With Flemish partners such as KU Leuven.
In addition to the Moonshot programs, RegMed XB actively conceives and facilitates other projects within the RegMed XB ecosystem. These initiatives are dedicated to technology platforms critical in the regenerative medicine field. For osteoarthritis: the LS-CarE project (2022-2027), funded by the Dutch Research Council (NWO).
-